The Carbonate Ion, Co32-, Can Best Be Described as Having:
Lewis Structure for COiii 2- | Carbonate ion
Lewis structure of carbonate ion is drawn in this tutorial step by step. Full valence electrons concept is used to draw the lewis structure of COiii ii-. Later on finishing the lewis structure of CO3 2-, at that place should be a -2 accuse and it should be stabile construction. You will larn about these facts in this tutorial.
Carbonate ion | CO3 2-
Carbonate ion has a -two accuse. Metal carbonate compounds are mutual in the world. Equally an CaCOiii can be given.
CO3 ii- Lewis structure
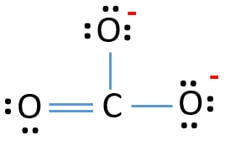
In that location are three σ bonds and π bond around carbon atom in the Lewis structure of CO3 2- ion.
Steps of drawing lewis structure of CO3 2-
Following steps are required to draw the CO3 2- lewis structure and they are explained in item in this tutorial.
- Discover total number of electrons of the valance shells of carbon and oxygen atoms
- Full electrons pairs
- Heart atom selection
- Put lonely pairs on atoms
- Check the stability and minimize charges on atoms by converting solitary pairs to bonds.
Drawing correct lewis structure is important to draw resonance structures of CO3 2- correctly.
Total number of electrons of the valance shells of COiii two-
Carbon is located at group 4 in the periodic tabular array. So, carbon has four electrons in its valence shell.Oxygen is located at 6th group. Information technology has six electrons in valence shell.
- Total valence electrons given by carbon atom = 4
There are three oxygen atoms in CO3 2- ion, Therefore
- Total valence electrons given by oxygen atoms = 6 *3 = xviii
There are -two accuse on CO3 2- ion. Therefore at that place are two more than electrons which contribute to the valence electrons.
- Full valence electrons = four + eighteen + 2 = 24
Full valence electrons pairs
Full valance electrons pairs = σ bonds + π bonds + alone pairs at valence shells
Total electron pairs are determined by dividing the number full valence electrons by two. For, COthree 2- ion, Full pairs of electrons are 12.
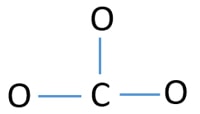
Center atom of CO3 2- ion
To exist the center atom, power of having higher valance is of import. Carbon has the more chance to be the center atom (Come across the figure) considering carbon can testify valance of iv. Oxygen's highest valence is 2. And then, now nosotros tin can build a sketch of COiii 2- ion.
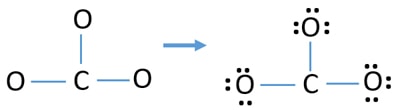
Lonely pairs on atoms
- There are already three C-O bonds in the higher up sketch. Therefore only nine (12-3 = 9) valence electrons pairs are remaining.
- First, marker those nine valence electrons pairs as solitary pairs in exterior atoms (on oxygen atoms). Ane oxygen cantlet volition have iii lone pairs following the octal rule (oxygen atom cannot continue more than eight electrons in its valence trounce).
- For iii oxygen atoms, nine electrons pairs are spent. Now there is no more alone pairs to marking on carbon atoms afterwards mark on oxygen atoms (exterior atoms).
Charges on atoms
Afterward, marking electron pairs on atoms, we should mark charges of each atom. Marker of charges are significant considering it is used to decide the best lewis structure of the ion. After marking charges, y'all volition run across, each oxygen atom will get a -1 accuse and carbon atom become a +1 charge.
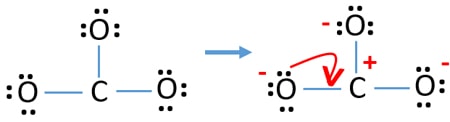
Check the stability and minimize charges on atoms past converting solitary pairs to bonds
- Oxygen atoms should concur negative charges considering electronegativity of oxygen cantlet is higher than carbon atom. Otherwise, we can say, power of belongings negative charges is greater in oxygen atoms than carbon atoms.
- The drawn structure is not a stable one because all atoms have a charge (Lewis construction should be a stable i with less charges).
- Now, we should try to minimize charges by converting alone pair or pairs to bonds. So catechumen i solitary pair of i oxygen cantlet to brand a C-O bond.
- At present there is a double bail between carbon atom and one oxygen atom (one C=O bond). Now, there are 2 unmarried bonds between carbon atom and other two oxygen atoms (two C-O bonds).
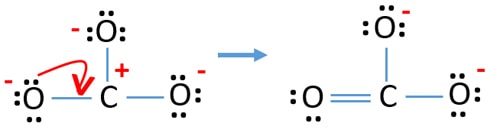
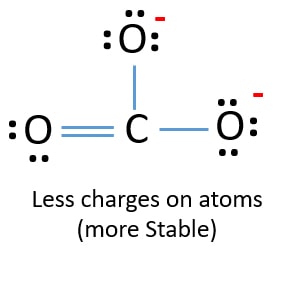
In new structure, charges of atoms are reduced than previous structure. Now there are no charge on on one oxygen atom and the carbon cantlet. Too, only two oxygen atoms accept -ane negative charges. At present you lot sympathize this structure of CO3 2- is more stable than previous structure. And so, this structure has more chance to be the lewis structure of COiii 2- ion.
Lewis structure of CO3 2- (carbonate) ion
Questions
carbonate lewis structure and bicarbonate lewis construction
In carbonate ion, in that location is ii oxygen atoms which has -1 charge on each of them. I of these oxygen atom take a proton (H+ ion) and form a -OH group.
Source: https://www.chemistryscl.com/general/lewis-structure-of-CO32-/
0 Response to "The Carbonate Ion, Co32-, Can Best Be Described as Having:"
ارسال یک نظر